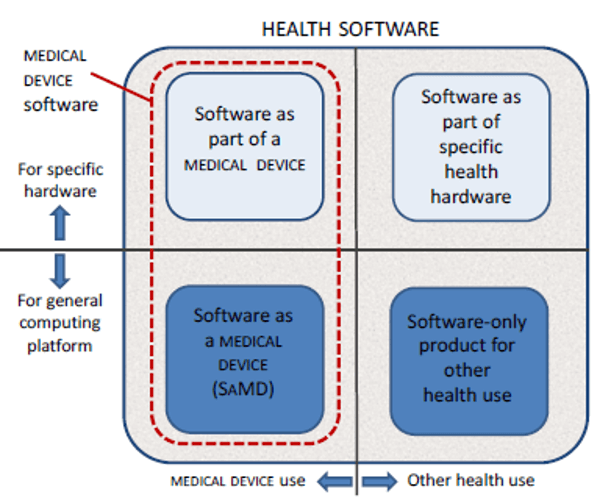
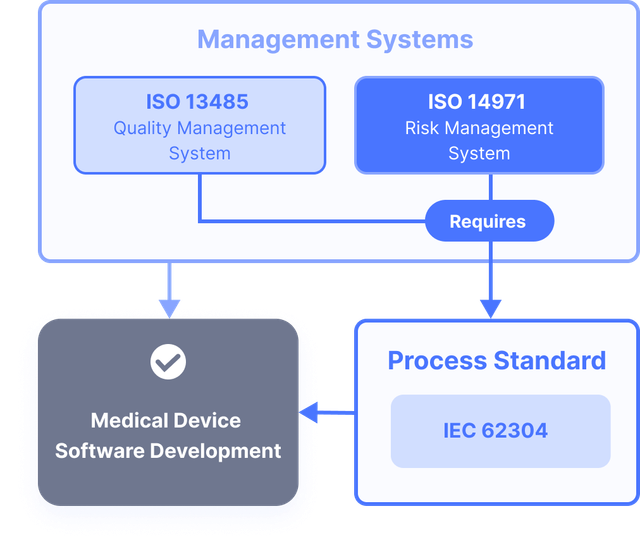
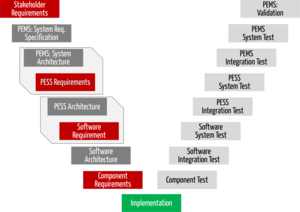
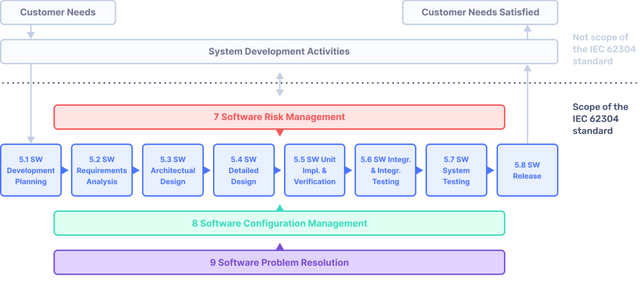
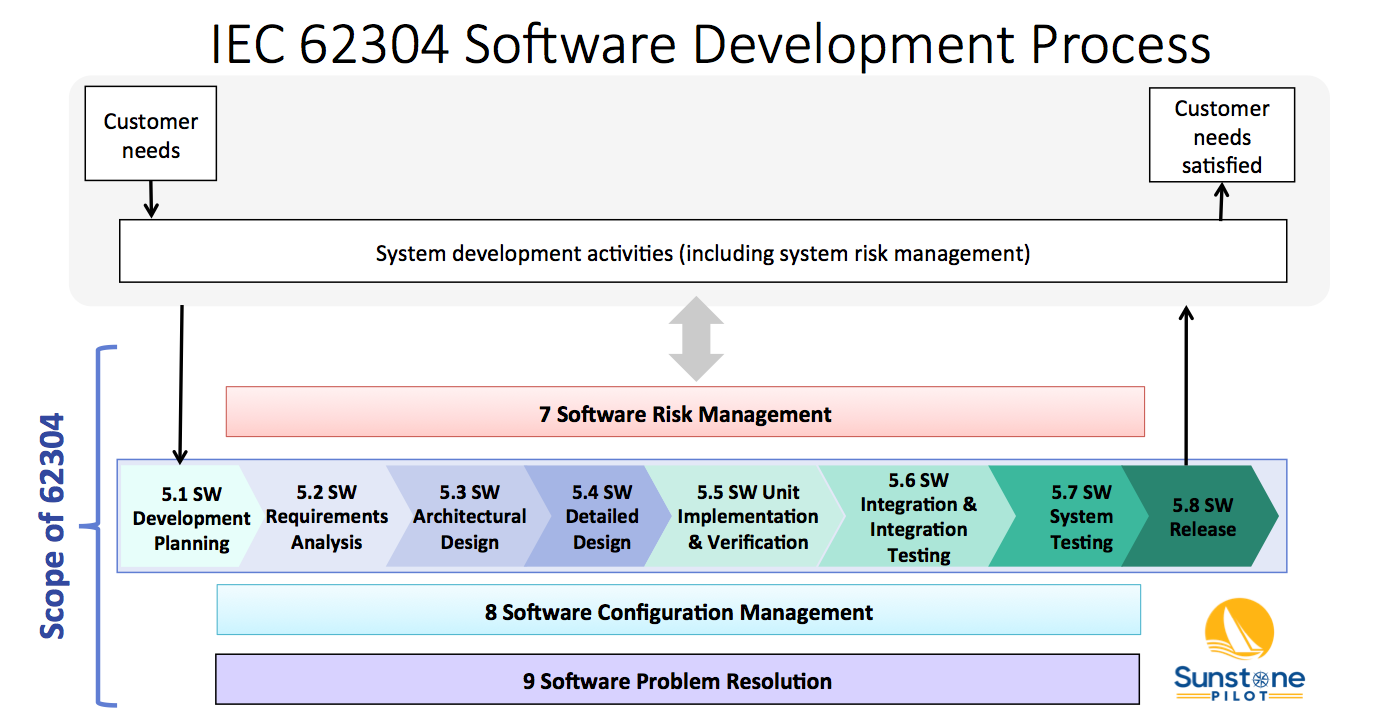
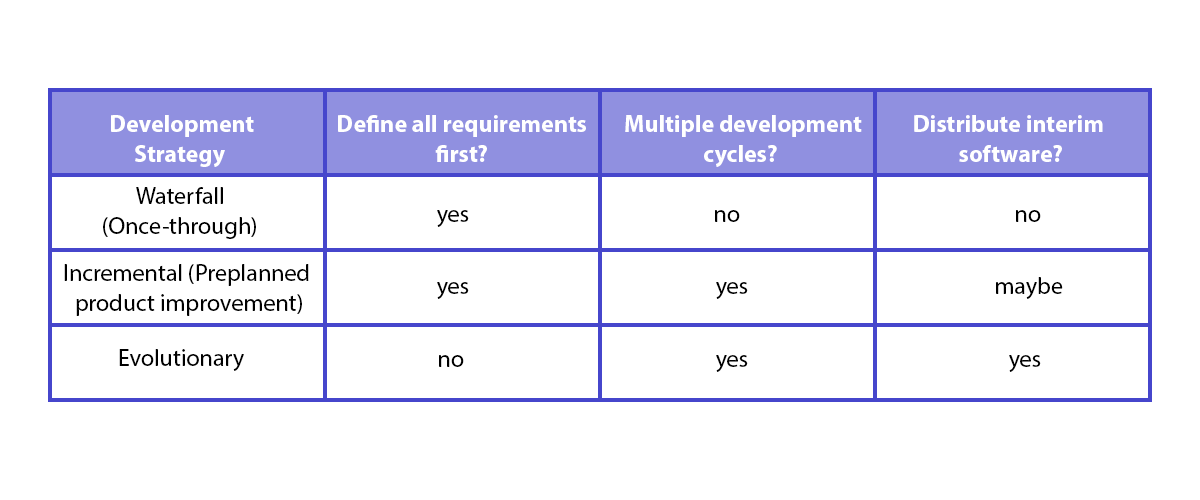
Implementing IEC 62304 for Safe and Effective Medical Device Software — PART 1 - Medical Design Briefs
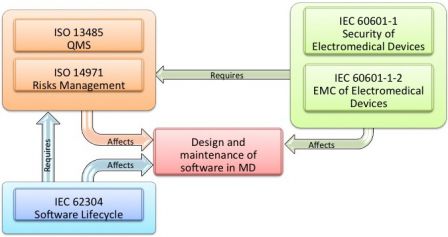
MD and IVD standards: IEC 60601-1 and IEC 61010-1, versus IEC 62304 - Part 1 - Software in Medical Devices, by MD101 Consulting
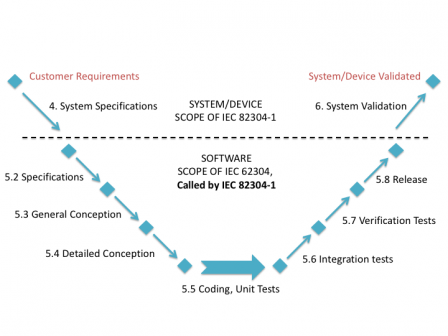
IEC 82304-1 - Consequences on agile software development processes - Software in Medical Devices, by MD101 Consulting